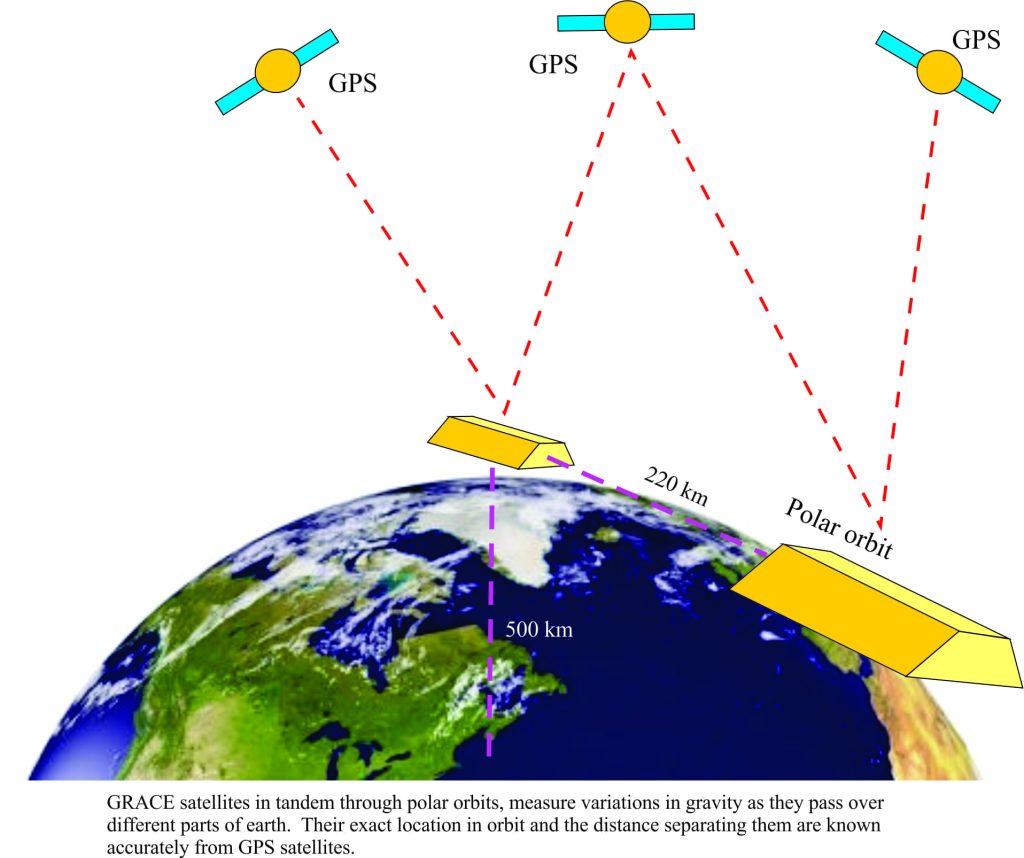
Monitoring groundwater from space
You could be excused for labeling this title conspiratorial, the brutal reality encapsulated in Alan Parsons prog rock group’s signature song or a Helen Mirren thriller. And if that is your inclination, don’t bother reading any further because this post deals with far more mundane uses of remotely sensed data. The data sets are generated by two groups of satellites that measure very different attributes of the earth, gravity and light spectra. Teasing the data has given us multiple stories of how systems like surface and groundwater are responding to human activities and natural processes. Some of these stories make grim reading.